 However, Avogadro’s idea was a hypothesis, and not a proven theory it was not universally accepted and much confusion remained, since variable and even multiple valences occurred for the bulk of the elements.īefore the periodic table could be formulated, true atomic weights needed to be determined, and accordingly it was necessary to know the correct valences for the elements. This recognition doubled (or even tripled) many of the atomic weights - in the case of water, giving hydrogen = 1 and oxygen = 16. Amedeo Avogadro (1776-1856) in 1821 had proposed that since two volumes of hydrogen gas unite with one volume of oxygen gas, then water is composed of two atoms of hydrogen and one atom of oxygen. In a previous chapter, we described how John Dalton, the founder of atomic theory, stated that the formula of water was HO, and that consequently the respective atomic weights of hydrogen and oxygen were 1 and 8. Frequently the true atomic weight of an element was not certain, but instead could be a multiple or fraction of the apparent equivalent weight. Unfortunately, even though Berzelius had made very precise measurements, the valence of the elements was not well understood. These arithmetic comparisons depended upon the accurate atomic weights determined by Berzelius ( discussed in a previous chapter). For example, for the alkaline earths, the atomic weight of strontium (42.5) was the arithmetic mean of calcium (20) and barium (65). Johan Wolfgang Döbereiner (1780-1849) in 1829 of the University of Jena (the same university where Karl Marx studied) noticed that there were several “triads” of chemically similar elements in which the middle element had an atomic weight half-way between those of the other two elements. However, scientists are forever searching for fundamental order and patterns, and as additional elements were discovered, trends were observed. 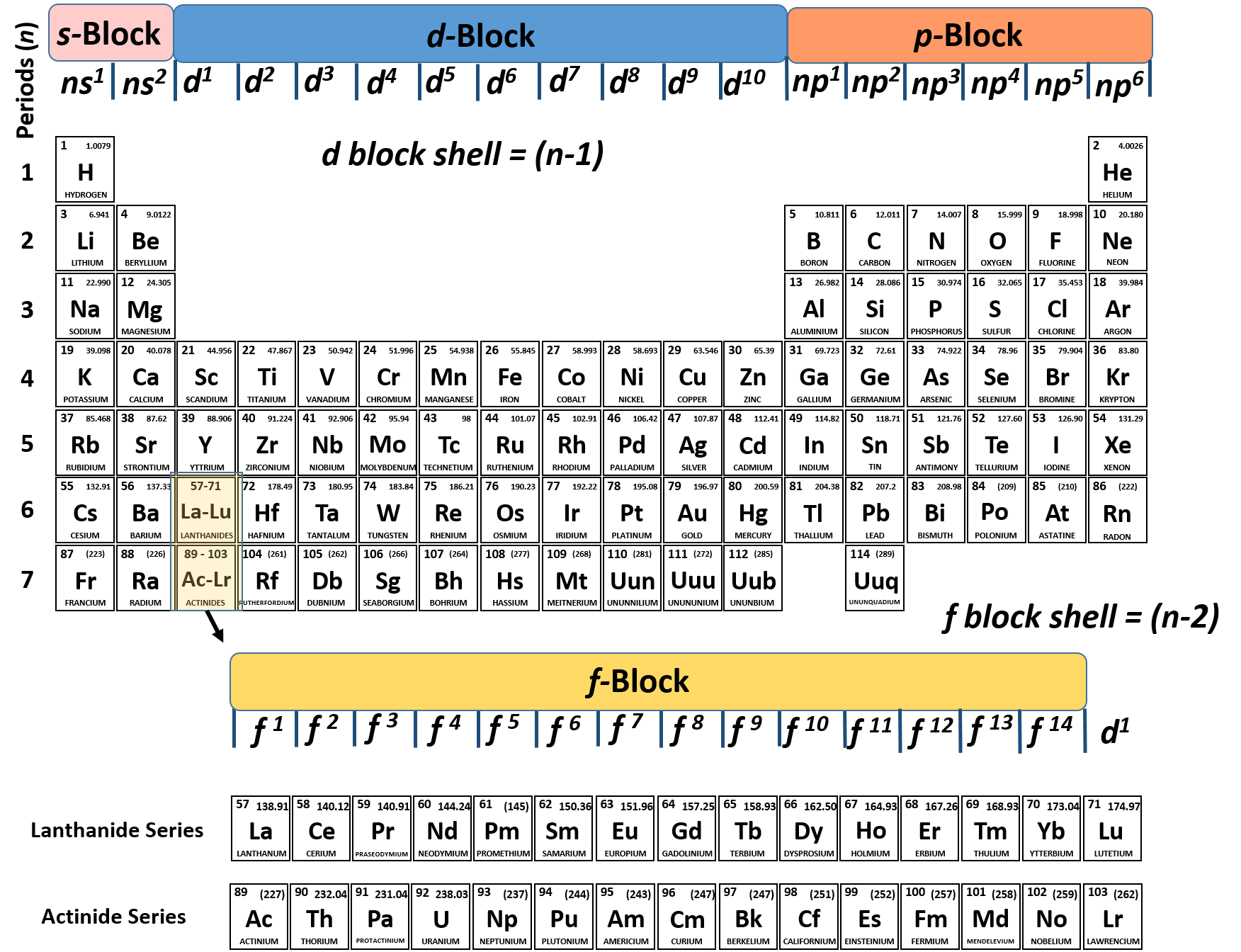
Lavoisier’s list was composed of a medley of “simple substances” (as he called them) which he organized into four categories: basic elements (gases), metals, nonmetals, and earths. 
It is very simple to use, although it comes packed with a comprehensive set of features.In 1789 Lavoisier turned the concept of “element” on its head when he proposed that water was a compound and that hydrogen, oxygen, carbon, sulfur, iron, copper, and 25 other substances were the true elements. The program uses a moderate amount of system resources, includes a well-drawn help file (with snapshots) and didn't cause freeze, crash or pop up errors during our tests. It's also possible to study the biography of famous persons who have made significant contributions to chemistry, as well as to access a glossary of all information provided by this application. Plus, you can view a graph of the atomic mass, search for text, view electron configuration and enhanced images of the elements, and much more. atomic mass, density), atomic properties (e.g. In addition, you can change the display mode to basic properties (e.g. You can also view data about alkali metals, transition metals, non metals, halogens, and others. Comprehensive details given for each elementįurthermore, if you click an element, you will be able to read detailed information about it, in a panel on the right side of the screen. 
The periodic table is automatically displayed and if you place the cursor over an element, you can view its picture, if one is available. The user interface of the program is plain and simple. Once you initiate it, you automatically receive a daily tip which explains the meaning of the current date in the chemistry world (if needed, this can be disabled from popping up). This software can come in handy if you are trying to learn the fundamentals of chemistry. Periodic Table is a software tool that displays the chemical elements found in the periodic table, as well as their image representations, where they are available. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
